Dr. Stijn Hinterding
Leonard S. Ornstein Laboratory, room 0.18
Princetonplein 1, 3584 CC Utrecht
P.O. Box 80 000, 3508 TA Utrecht
The Netherlands
phone: +31 (0)30 253 2315
secretariat: +31 (0)30 253 2952
e-mail: s.o.m.hinterding@uu.nl
Supervisor: dr. Freddy Rabouw
Research
Supervisor: Dr. Freddy Rabouw
Promotors: Prof. dr. ir. Bert Weckhuysen and Prof. dr. Alfons van Blaaderen
Funding: MCEC
Employed: 1 November 2016 – 30 October 2020
Studying single catalytic conversion events using fluorescence correlation spectroscopy
To unravel the heterogeneous nature of an ensemble of catalyst particles, it is necessary to study individual nanoparticles. So far, one approach has been the study of single nanoparticles catalyzing the formation of fluorescent molecules from organic precursors. By collecting photons emitted by the fluorescent product on a CCD camera, it is possible to map catalytic events with a high spatial resolution (< 100 nm).[1]–[3] In this way changes in catalytic activity of single nanoparticles over time, as well as differences in activity between regions in porous catalyst particles have been observed.[1]–[3] Although these methods provide great insight into catalytic activity on the single particle level, their temporal resolution is quite low, on the order of 10 ms. As such, these methods cannot provide detailed insight into fast phenomena, such as diffusion and adsorption.
We aim to gain new insight into catalysis on the single particle and single molecule level, by studying single catalytic events with high temporal resolution. To this end, the conversion of one dye molecule into a differently coloured dye molecule, over a single catalyst particle, will be used as a model system. Subsequently, we can obtain information on conversion events by detecting single photons emitted by the reactant and product. By using detectors with high detection efficiency and temporal resolution (~100 ps), we can apply the method of fluorescence correlation spectroscopy to gain insight into reactant and product diffusion, adsorption, and differences in catalytic activity in space and time.
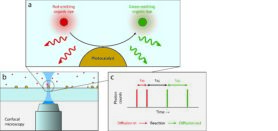
Figure 1. (a) Fluorescent molecules are converted into differently coloured molecules by a photocatalyst. (b) By analyzing the system using a microscope and detectors with high temporal resolution, (c) we will be able to detect single photons emitted by the reactant (red) and the product (green). By correlating these photons, we hope to gain insight into the diffusion of the reactant and product, as well as the reaction dynamics.
[1] Y. Zhang et al., Proc. Natl. Acad. Sci. U.S.A. 112, 29, 8959–8964 (2015)
[2] Z. Ristanovic et al., Angew. Chemie – Int. Ed. 54, 1836–1840 (2015)
[3] J. B. Sambur et al., Nature 530, 77–80 (2016)